 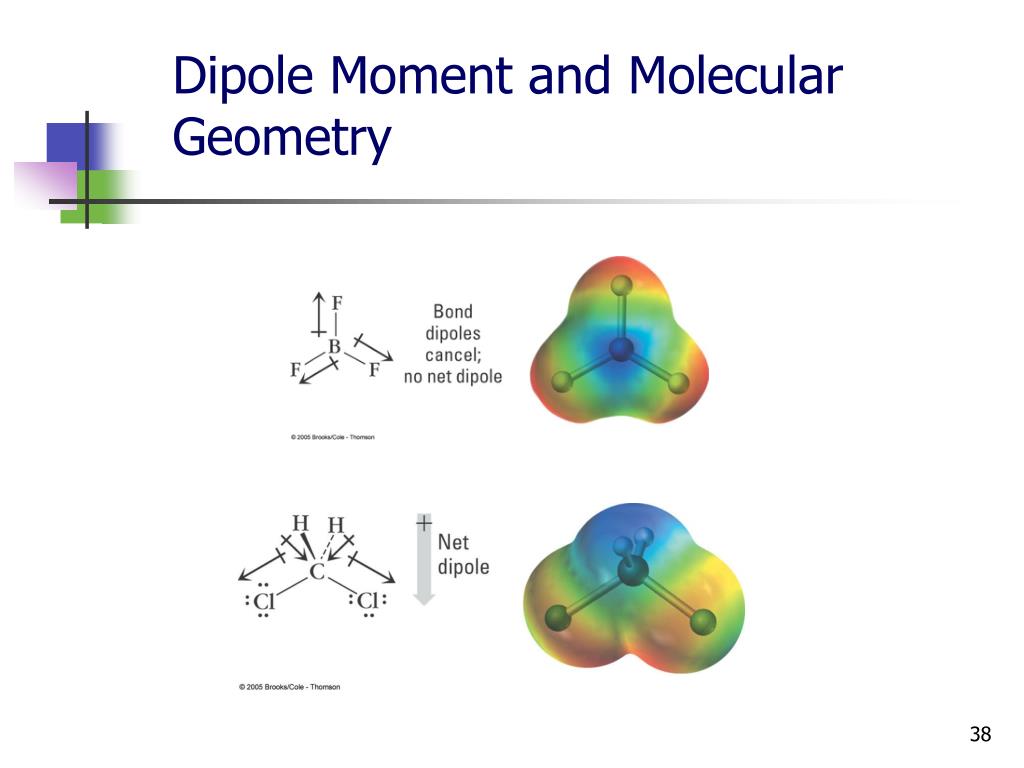
As the shape of the molecule is symmetric with even charge distribution, it makes Phosphorus Pentafluoride a nonpolar molecule.Trending Questions How big was Pillotex before it went bankrupt? To circle around an object in space? Why did they downgrade Pluto? What is attacking earth and blowing up structures? How far away is dorset? Do mainstream astronomers recognize the existence of Nibiru? Why does a probe need scientific instruments? How long does nightfall-twilight last? How can you tell how far away a strom is? How many cultures or civilizations use equinoxes or solstices in their life? What is meteorite bombardment? How do scienctists determine how long meteorites have been in space? Which military communications satellite is the most advanced to date and consist of 5 satellites in geosynchronous orbits? How do you treat a Globular Maxillary cyst? What are three reasons an astronomers work is important? Why do we sometimes see only part of the near side of the moon? How did continuous bombardment of meteorites and asteroids affect the temperature and size of earth? What planet in our solar system day is more than half as long as its year? What type of elements are usually found in meteorites? For most of history Euerepeans belived that the sun plantes and stThis began to change in the 1500s when a polish astronomer offered his theory that earth revolves around the sun. PF 5 is a nonpolar molecule because there is no net dipole moment in the molecule. Which of the following molecules has (have) a non-zero dipole moment: SF4, SiF4, XeF4 a. But here as the PF5 molecule doesn’t have any poles, the charges are distributed evenly in the molecule. The molecules that have net dipole moment have regions of partial positive charges and partial negative charges. As a result, there is none or zero net dipole moment in PF 5. To understand the polarity of this molecule one needs to know the shape and lewis structure, net dipole moment and the. 
This molecule is used for polymerization catalysts. The molecule has five single bonds between each Fluorine and Phosphorus atoms. All these dipole moments will be nullified because of the symmetric distribution of the atoms in the molecule. Phosphorus Pentafluoride, having a chemical formula of PF5 is a colourless gas. But this dipole moment will be from the Phosphorus atom towards the Fluorine atom. It has one F on top and the other three as a base for a tetrahedral structure. Some molecules with a permanent dipole moment include SF4 and XeF4, while others like SiF4 and BF3 are non-polar. Phosphorus atoms has an electronegativity value of 2.19 and Fluorine’s electronegativity value is 3.98 The difference of electronegativities for both these atoms is quite higher than 0.4 which means that there can be a dipole moment in the molecule. The factors affecting the dipole moment include electronegativity difference, molecular geometry, and bond polarity. Generally, the molecules having a symmetric distribution or arrangement are nonpolar in nature. A: To solve this question, the hybridization of nitrogen and structural aspects such as delocalization. The arrangement of the atoms is quite symmetric for this molecule. For many characters, the only way to incorporate an Ultra into a larger combo is to use an FADC to make it possible. Often, they're used to make otherwise-unsafe moves safe, or to extend combos. The molecular geometry of this molecule is Trigonal Bipyramidal, which means two atoms of Fluorine are located in an axial position, and the other three are in the equatorial position. Click hereto get an answer to your question Which of the following molecule/molecules will be have zero dipole moment H2O, CO2, CCl4, CHCl3, NH3, BF3,BeF2. 6 Many moves in Street Fighter IV can be FADC'ed, and it's a cornerstone of higher-level gameplay. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
